Thomson's Plum Pudding Model
Key Points:
1) JJ Thomson used the cathode ray experiment to prove
- Atoms are made of smaller parts (can be split)
- electrons are small negative particles
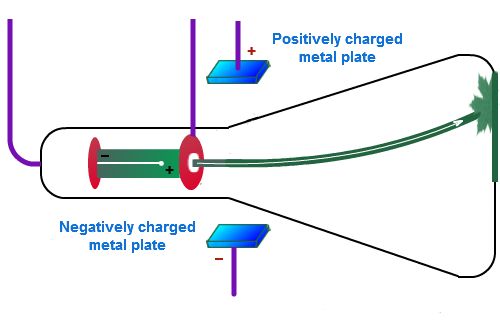
Cathode Ray Tube ExperimentThomson experimented with a cathode-ray tube. The tube is nearly empty, but shoots a green beam across it when electricity is sent through it.
He discovered that a positively charged plate (on top) attracted the beam and made it curve up. Thomson concluded that the beam must be made of tiny particles with a negative charge, because opposite charges attract. That negative particle Thomson discovered is now called the electron. |
Plum Pudding Model
|
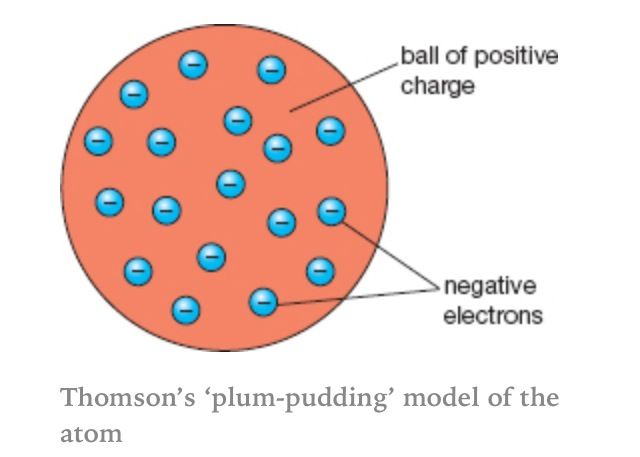
Dalton's atom had nothing in it so, Thomson had to update the atomic model to include electrons. His experiment proved electrons, but not their location in the atom. He said the electrons are spread out in the atom, and are in a positive ball of charge. He said this because atoms have no charge so the positive part must equal the negative electrons.
He called it the plum pudding model. The plums are the electrons, and the cake (or pudding) is the positive ball. |