Rutherford's Nuclear Model
Key Points
- Ernst Rutherford used the Gold Foil Experiment to discover and prove:
- The solid positive nucleus of an atom
- The atom is mostly empty space
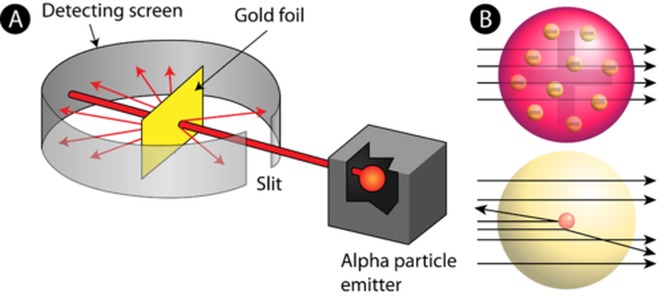
Gold Foil Experiment
Figure A (left): The set up of the experiment.
Figure B (right): The expectations of different atomic models.
Figure B (right): The expectations of different atomic models.
|
In 1909, a former student of Thomson’s named Ernest Rutherford decided to test Thomson’s Plum Pudding Model. He designed the Gold foil experiment to show if atoms had different parts. He aimed a beam of small, positively charged particles at a thin sheet of gold foil. Rutherford put a special detecting coating behind the foil. The coating glowed when hit by the positively charged particles. Rutherford could then see where the particles went after hitting the gold. This experiment would show if atoms have different parts or if they are all the same throughout, as the plum-pudding model suggested. (Figure A)
Rutherford started with Thomson’s idea that atoms are soft “blobs” of matter through which electrons are evenly distributed. Therefore, he expected the particles to pass right through the gold in a straight line. (Figure B) Most of the particles went straight through. But surprisingly, a few of the particles were deflected (turned to one side). Some even bounced straight back! |
It was quite the most incredible event that has ever happened to me in my life. It was almost as if you shot a fifteen inch shell into a piece of tissue paper and it came back to hit you! |
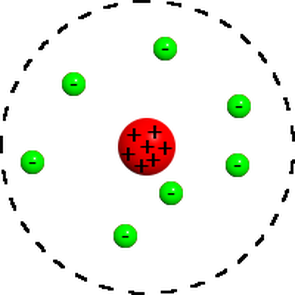
Nuclear Model
|
The Plum Pudding Model did not explain the results to the gold foil experiment If Thomson was right all the particles would go straight through. (Figure B, above).
To explain his experiment, Rutherford, stated that atoms have a very small, very dense, positive center called a nucleus. the reason it is dense and positive and small is because most positive particles went straight through (so it is small). But some positive particles bounced backward, so it must be heavy and positive (likes repel). The Nuclear Model has a small, dense, and positive nucleus, with electrons surrounding it. Both the positive charge and the negative charge are equal in a neutral atom. |