Schrodinger and Heisenberg Model
Key Points:
- Using mathematics Schrödinger and Heisenberg developed their theory
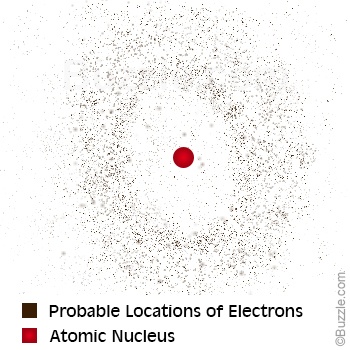
- The electrons are in likely the regions called clouds, not single regions.
Theory and Model
|
An Austrian physicist named Erwin Schrödinger and a German physicist named Werner Heisenberg added a more confusing but important part to the model of the atom. They based their theory on mathematics, and not experiments. They said you can't know the exact location of an atom and that electrons may not be particles but waves.
They said that electrons do not stay in one nice ring orbit. In fact, their model states that you can't know exactly where the electron is at anytime. So instead of rings, there are regions inside the atom where electrons are most likely to be found. These regions are called electron clouds, or orbitals. The electron-cloud model of the atom is shown to the late. |