Bohr's Model of the Atom
Key Points
The key points for this webpage are:
Bohr used spectroscopy (hydrogen light) to find:
Bohr used spectroscopy (hydrogen light) to find:
- Electrons only travel in certain places, called (orbits,shells)
- Bohr's Model has a certain way to place electrons.
Spectrum Experiments
|
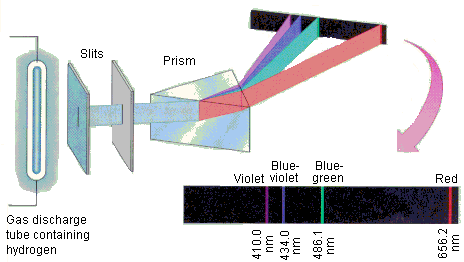
In 1913, Niels Bohr, a Danish scientist who worked with Rutherford, studied the way that atoms react to light. He used a technique called spectroscopy to look at the way light was split only into certain colors for each element. It required a light like a neon light to be shined through a prism and the wavelength of light to be measured. Hydrogen is shown to the left.
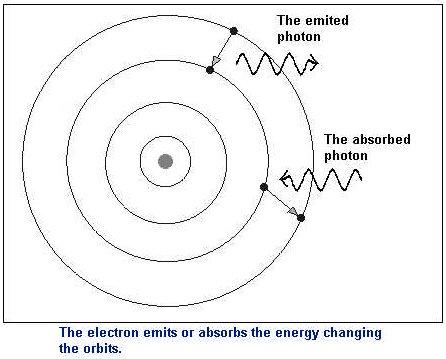
Bohr’s results led him to propose that electrons move around the nucleus in definite paths, also called levels, shells, or orbits. In Bohr’s model, there is nothing in between the levels, the electrons can only be in one level or another, but not between. The electrons can absorb energy and jump up to a higher level. Think of the levels as rungs on a ladder. You can stand on the rungs of a ladder but not between the rungs. The electrons can also lose energy and fall down a level (by releasing energy as light). This is shown on the left. Bohr’s model was a valuable tool in predicting some atomic behavior/ |
Bohr Model
|
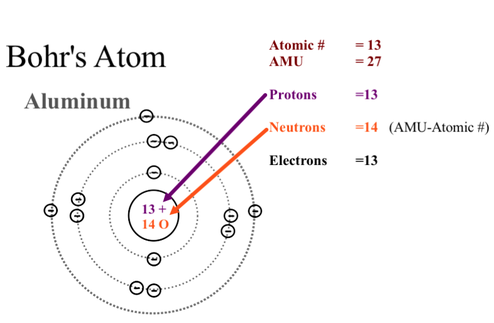
The Bohr Model has electron shells depending on the number of electrons. Each shell has its own maximum number that it can hold. Shell Number Maximum Electrons (for us)
1 2 2 8 3 8 4 2 There is also an order to fill in the shells, and make a Bohr Diagram.
|